• Specific advantages and disadvantages
The advantages of MF are:
• Low operating pressure required;
• Low energy consumption for semi dead-end set-up, compared to nano-filtration or reverse osmosis;
• Few manual actions required;
• Relatively cheap;
• No energy-consuming phase transfer needed, such as e.g. evaporation techniques;
• Quality of the produced permeate is not determined by the management.
• Possible disadvantages of MF are:
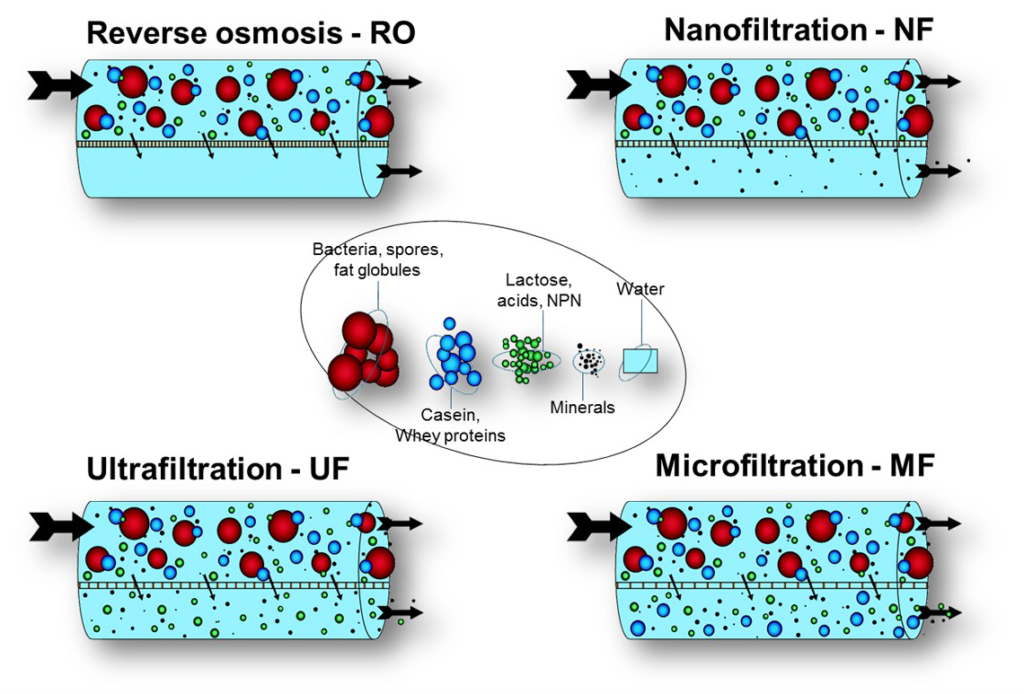
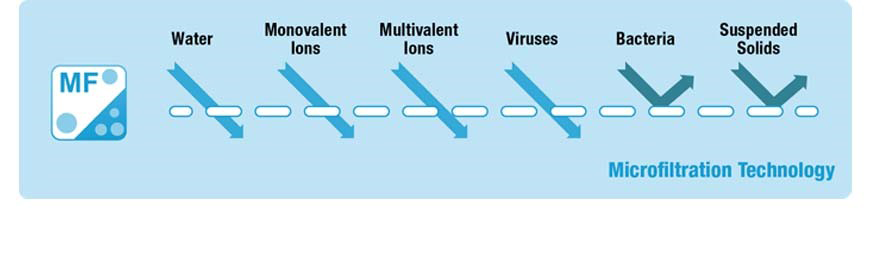
• Only suspended matter and bacteria removed ((~log 5 removal);
• Sensitive to oxidative chemicals (e.g. nitric acid, sulphuric acid, peroxide and persulphate in high concentrations);
• Damage can be caused by hard and sharp particles > 0.1 mm, whereby pre-filtration is necessary;
• Membrane damage if re-rinsed at pressure in excess of 1 bar.
• Application
Micro-filtration is primarily used as a pre-treatment step in the production of drinking and process water. It has excellent properties for removing suspended matter, bacteria and cysts. It is an alternative to classic sand filtration. Further, cross-flow MF is used in the:
• Dairy industry (cheese, milk,…);
• Food industry (clarification of fruit juice, wine, beer, etc.);
• Metal industry (oil/water emulsion separation);
• Textile industry (effluent treatment);
• Pharmaceutical industry (cold sterilisation).
MF is also implemented for the re-use of wastewater. In this case, it can be combined with a biological step in a membrane bioreactor.
• Boundary conditions
Membranes must be protected against hard particles larger than 0.1 mm. They can be removed by regular pre-filtration. Further, supply flows and pH conditions must be compatible with the membrane material.
• Effectiveness
MF can be implemented for removing the following parameters:
• Suspended matter (>99%);
• Harmful micro-organisms (e.g. bacteria, protozoa, algae, fungi) (>99%).
• PAHs.
Further, MF can also be implemented to break down emulsions.
• Support aids
Support aids like bleach, peroxide, acid, alkali or detergent can be used to chemically clean the MF installation.
• Environmental issues
The concentrate from an MF has a high concentration of suspended matter and bacteria. This can be discharged together with wastewater if discharge norms are not breached. Rinse waters after chemical cleaning contain substances like bleach and formed AOX, peroxide, acid and alkali. These rinse waters can only be discharged to specific waste purification systems.