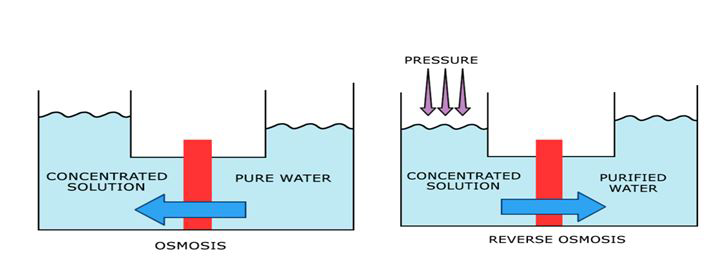
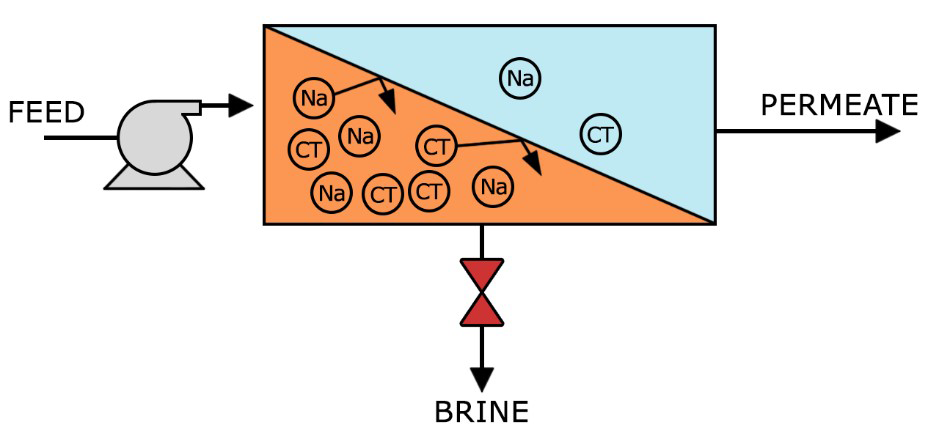
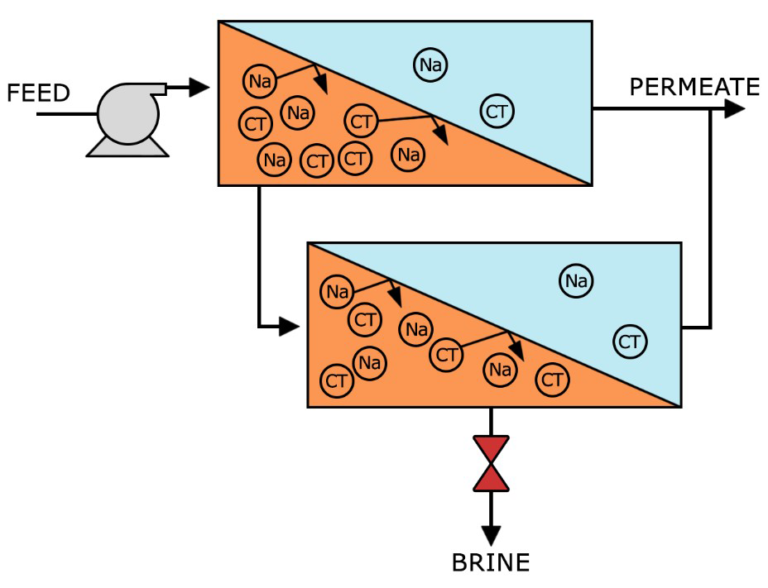
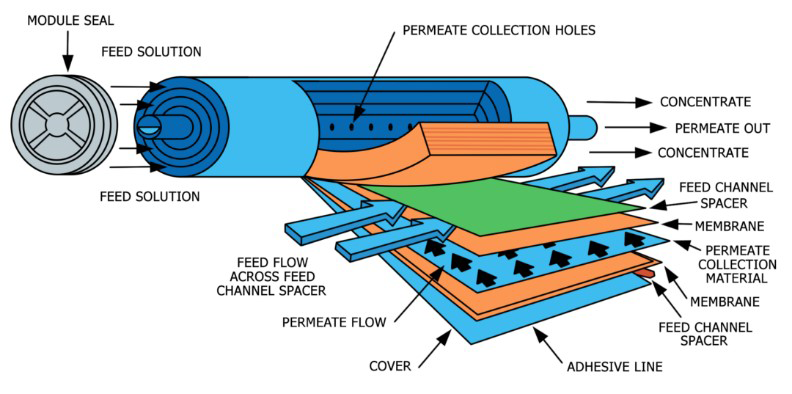
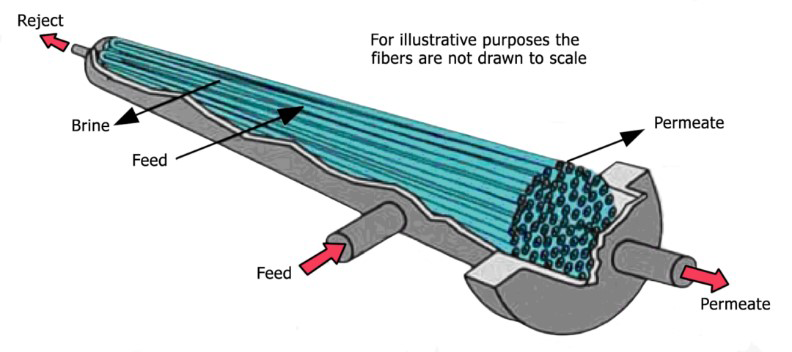
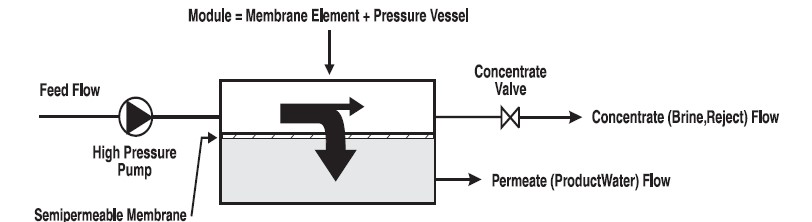
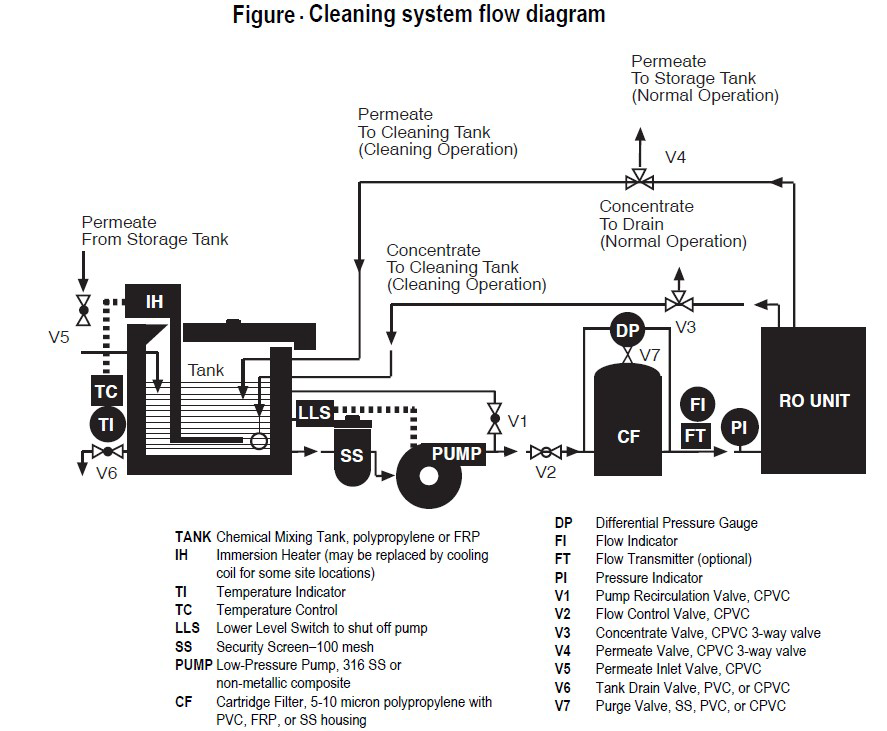
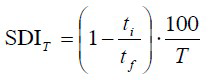With a high pressure pump, feed water is continuously pumped at elevated pressure to the membrane system. Within the membrane system, the feed water will be split into a low-saline and/or purified product, called permeate, and a high saline or concentrated brine, called concentrate or reject. A flow regulating valve, called a concentrate valve, controls the percentage of feedwater that is going to the concentrate stream and the permeate which will be obtained from the feed.
The key terms used in the reverse osmosis / nanofiltration process are defined as follows.
Recovery – the percentage of membrane system feedwater that emerges from the system as product water or “permeate”. Membrane system design is based on expected feedwater quality and recovery is defined through initial adjustment of valves on the concentrate stream. Recovery is often fixed at the highest level that maximizes permeate flow while preventing precipitation of super-saturated salts within the membrane system.
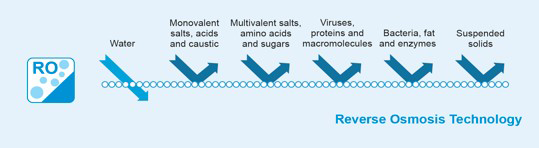
Rejection – the percentage of solute concentration removed from system feedwater by the membrane. In reverse osmosis, a high rejection of total dissolved solids (TDS) is important, while in nanofiltration the solutes of interest are specific, e.g. low rejection for hardness and high rejection for organic matter.
Passage – the opposite of “rejection”, passage is the percentage of dissolved constituents (contaminants) in the feedwater allowed to pass through the membrane.
Permeate – the purified product water produced by a membrane system.
Flow – Feed flow is the rate of feedwater introduced to the membrane element or membrane system, usually measured in gallons per minute (gpm) or cubic meters per hour (m3/h). Concentrate flow is the rate of flow of non-permeated feedwater that exits the membrane element or membrane system. This concentrate contains most of the dissolved constituents originally carried into the element or into the system from the feed source. It is usually measured in gallons per minute (gpm) or cubic meters per hour (m3/h).
Flux – the rate of permeate transported per unit of membrane area, usually measured in gallons per square foot per day (gfd) or liters per square meter and hour (l/m2h).
Factors Affecting Reverse Osmosis and Nanofiltration Performance
Permeate flux and salt rejection are the key performance parameters of a reverse osmosis or a nanofiltration process. Under specific reference conditions, flux and rejection are intrinsic properties of membrane performance. The flux and rejection of a membrane system are mainly influenced by variable parameters including:
– pressure
– temperature
– recovery
– feed water salt concentration
PRETREATMENT
Processes that rely on microporous membranes must be protected from fouling. Membrane foul-ing causes a loss of water production (flux), reduced permeate quality, and increased trans-membrane pressure drop.
Membrane fouling is typically caused by precipitation of inorganic salts, particulates of metal oxides, colloidal silt, and the accumulation or growth of microbiological organisms on the membrane surface. These fouling problems can lead to serious damage and necessitate more frequent replacement of membranes.
Water Chemistry and Pretreatment
To increase the efficiency and life of reverse osmosis (RO) systems, effective pretreatment of the feed water is required. Selection of the proper pretreatment will maximize efficiency and membrane life by minimizing:
– Fouling
– Scaling
– Membrane degradation
Optimizing:
– Product flow
– Product quality (salt rejection)
– Product recovery
– Operating & maintenance costs
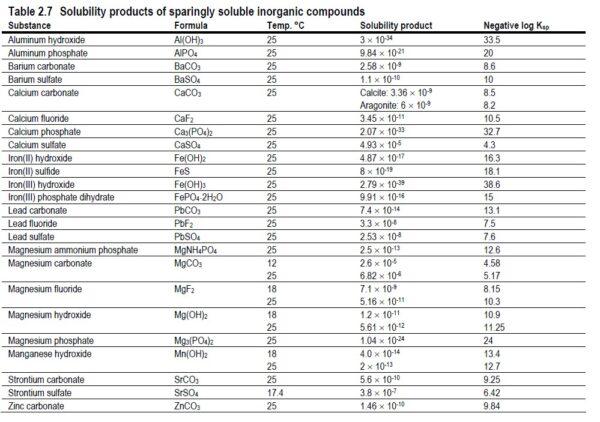
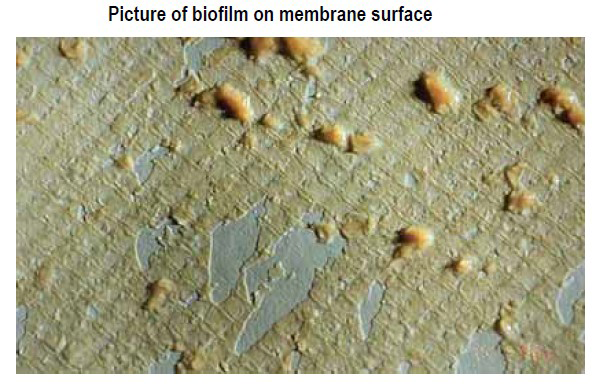
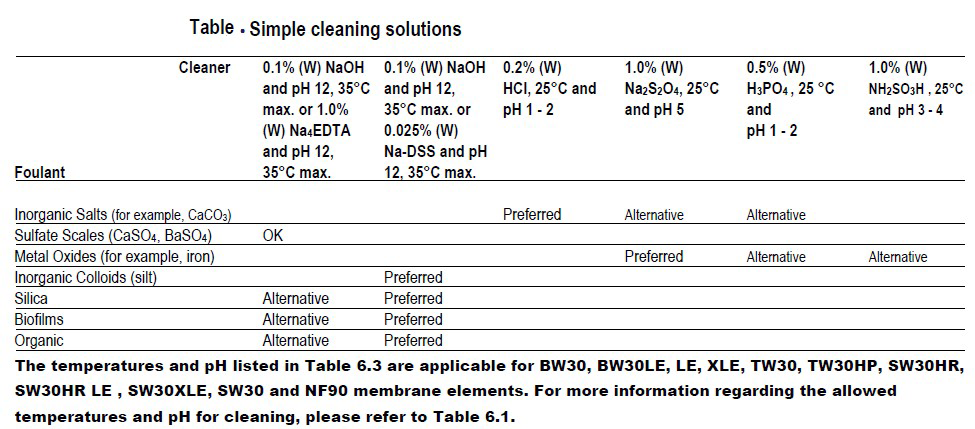
Fouling is the accumulation of foreign materials from feed water on the active membrane surface and/or on the feed spacer to the point of causing operational problems. The term fouling includes the accumulation of all kinds of layers on the membrane and feed spacer surface, including scaling. More specifically, colloidal fouling refers to the entrapment of particulate or colloidal matter such as iron flocs or silt, biological fouling (biofouling) is the growth of a biofilm, and organic fouling is the adsorption of specific organic compounds such as humid substances and oil on to the membrane surface. Scaling refers to the precipitation and deposition within the system of sparingly soluble salts including calcium carbonate, barium sulfate, calcium sulfate, strontium sulfate and calcium fluoride.
Pretreatment of feed water must involve a total system approach for continuous and reliable operation. Such inadequate pretreatment often necessitates frequent cleaning of the membrane elements to restore productivity and salt rejection. The cost of cleaning, downtime and lost system performance can be significant.
The proper treatment scheme for feed water depends on:
– Feed water source
– Feed water composition
– Application
Summary of Pretreatment Options
Summarizes the pretreatment options when specific risks for scaling and fouling are present. It is a quick reference for “possible” and “very effective” methods. A combination of “possible” methods may also be “very effective”.
Guidelines for Acceptable RO Feed Water
Below Table summarizes the limits of quality parameters of the feed water. It is recommended to respect these limits to ensure successful operation of the membrane system. Otherwise, more frequent cleaning and/or sanitization may become necessary. The concentrations correspond to the entry to the membrane for a continuous feed stream, including any influences to the feed water from dosing chemicals or piping materials in the pretreatment line.
Comments & conditions Max. level Unit Component
5 1 SDI
0.5 NTU Turbidity
Target: <1 4 1 MFI0.45
0.1 mg/L Oil & grease
Synthetic organic compounds (SOC) have generally more adverse effects on RO/NF membranes compared with natural organic matters (NOM). 3 mg/L TOC
10 mg/L COD
Target: <5 10 μg/l Ac-C AOC
Target: <1 5 pg/cm2 ATP BFR
Under certain conditions, the presence of chlorine and other oxidizing agents will cause premature membrane failure. Since oxidation is not covered under warranty, FilmTec recommends removing residual free chlorine by pretreatment prior to membrane exposure. 0.1 mg/L Free chlorine
pH <6, oxygen <0.5 ppm 4 mg/L Ferrous iron
0.05 mg/L Ferric iron
0.05 mg/L Manganese
0.05 mg/L Aluminum
Higher concentrations may damage the element glue line In ug/L range mg/L VOC’s
SOLID REDUCTION
Membrane feedwater should be relatively free from colloidal particulates. The most common particulates encountered in industrial membrane systems are silt, iron oxides, and manganese oxides.
Silt Density Index (SDI) testing should be used to confirm sufficient water quality for the specific membrane system employed. SDI evaluates the potential of feedwater to foul a 0.45 µm filter. Unacceptable SDI measurements can be produced even when water quality is relatively high by most industrial water treatment standards. Where pretreatment is inadequate or ineffective, chemical dispersants may be used to permit operation at higher-than-recommended SDI values. RO systems are highly susceptible to particulate fouling, EDI systems are more forgiving, and UF systems are designed to handle dirty waters.
Media Filtration
The removal of suspended and colloidal particles by media filtration is based on their deposition on the surface of filter grains while the water flows through a bed of these grains (filter media). The quality of the filtrate depends on the size, surface charge, and geometry of both suspended solids and filter media, as well as on the water analysis and operational parameters. With a well-designed and operated filter, a SDI15 <5 can usually be achieved.
Oxidation–Filtration
Some well waters, usually brackish waters, are in a reduced state. Typically, such waters contain divalent iron and manganese, sometimes hydrogen sulfide and ammonium, but no oxygen; therefore, they are also called anoxic. Often the oxygen has been used up (e.g., by microbiological processes) because the water is contaminated with biodegradable organic substances, or the water is from a very old aquifer.
In-Line Filtration
The efficiency of media filtration to reduce the SDI value can be markedly improved if the colloids in the raw water are coagulated and/or flocculated prior to filtration. In-line filtration can be applied to raw waters with a SDI only slightly above 5. The optimization of the method, also named in-line coagulation or in-line coagulation-flocculation, is described in ASTM D 4188 /25/. A coagulant is injected into the raw water stream, effectively mixed, and the formed microflocs are immediately removed by media filtration.
Coagulation-Flocculation
For raw waters containing high concentrations of suspended matter resulting in a high SDI, the classic coagulation-flocculation process is preferred. The hydroxide flocs are allowed to grow and settle in specifically designed reaction chambers. The hydroxide sludge is removed, and the supernatant water is further treated by media filtration.
For the coagulation-flocculation process, either a solids-contact type clarifier or a compact coagulation-flocculation reactor may be used. For details, please refer to the general water treatment textbooks.
Microfiltration/Ultrafiltration
Microfiltration (MF) or ultrafiltration (UF) membrane removes virtually all suspended matter and, in the case of ultrafiltration, also dissolved organic compounds depending on their molecular mass and on the molecular mass cut-off of the membrane. Hence, an SDI <1 can be achieved with a well-designed and properly maintained MF or UF system.
Cartridge Microfiltration
A cartridge filter with an absolute pore size of less than 10 μm is the suggested minimum pretreatment required for every RO system. It is a safety device to protect the membranes and the high pressure pump from suspended particles. Usually it is the last step of a pretreatment sequence. A pore size of 5 μm absolute is recommended. The better the prefiltration the less RO membrane cleaning required. If there is a risk of fouling with colloidal silica or with metal silicates, cartridge filtration with 1 to 3 μm absolute pore size is recommended. The filter should be sized on a flow rate according to the manufacturer’s recommendation and replaced before the pressure drop has increased to the permitted limit, but at least every 3 months.